 
The entropy of mixing may be calculated by Gibbs' Theorem which states that when two different substances mix, the entropy increase upon mixing is equal to the entropy increase that would occur if the two substances were to expand alone into the mixing volume. If the substances being mixed are initially at different temperatures and pressures, there will, of course, be an additional entropy increase in the mixed substance due to these differences being equilibrated, but if the substances being mixed are initially at the same temperature and pressure, the entropy increase will be entirely due to the entropy of mixing. 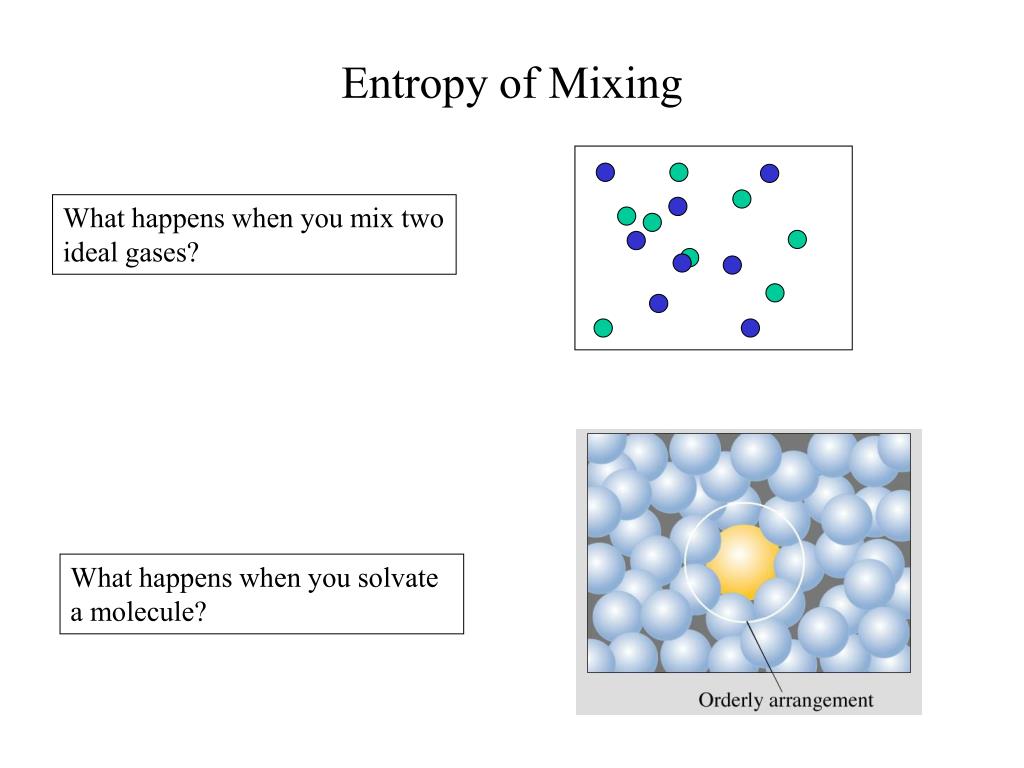
We assume that the mixing process has reached thermodynamic equilibrium so that the mixture is uniform and homogeneous. This entropy change must be positive since there is more uncertainty about the spatial locations of the different kinds of molecules when they are interspersed. This is not a standard procedure and is not useful except that it does demonstrate very clearly the role of mixing in helping to determine the position of chemical equilibria.The entropy of mixing (also known as configurational entropy) is the change in the entropy, an extensive thermodynamic quantity, when two different chemical substances or components are mixed. It is interesting to use equation (E3) to derive the relation between D G and the equilibrium constant K. The decrease in free energy on mixing is always a strong force promoting solubility. For the ideal solution, as defined above, the Gibbs free energy from equation (E3) is always negative and becomes more negative as the temperature is increased (see diagram). 
Its variation with concentration is shown in the diagram below. Since the mole fractions in equation (E2) are always less than unity, the ln terms are always negative, and the entropy of mixing is always positive. This immediately leads to the result that the Gibbs free energy of mixing is It is one for which the entropy of mixing is given by equation (E2) and the enthalpy of mixing is zero. We now give a possible definition of an ideal solution. Although equation (E1) cannot in any sense be proved, it is one of the cornerstones of models of mixing of liquids. There are other ways of justifying equation (E1) based on lattice models, which have different merits and different drawbacks. It has only been given to provide a justification of equation (E1). This does not occur when the differences are very large, as in polymer solutions (see Third Year Lecture Course on Amphiphiles and Polymers), and it would also not be wise to scrutinize too hard the details of the derivation above. This indicates that the two assumptions made in the derivation above largely cancel each other. We then derive an expression for the entropy of mixing of two liquids as shown in the panel below.Įxperimentally, equation (E2) is found to account surprisingly well for the entropy of mixing for molecules that only interact via van der Waals forces, even when the sizes of the two species differ by factors of two or three. 
We now assume that we can apply equation (E1) to liquid mixing but with the volumes revised to take account of the actual available volumes. The fraction of volume available in liquid B for molecules of A to expand into is n Bf BV B where n B is the number of molecules of B, f B is the fraction of the molecular volume that is not occupied by the molecule itself, and V B is the volume occupied by one molecule of B in the liquid. The B molecules themselves occupy the major part of the total volume of liquid B. When liquid A expands into the space occupied by B it does not have access to all the volume It is obviously not possible to apply equation (E1) directly to the mixing of liquids. By calculating the entropy of expansion of each gas we can calculate the entropy of mixing as shown in the panel below. Thus, for gas A the available volume has increased from V A to ( V A + V B). Since the molecules of ideal gases do not interact the increase in entropy must simply result from the extra volume available to each gas on mixing. For the mixing of two ideal gases it is straightforward to obtain an expression for the entropy of mixing. This is qualitatively easily visualised in terms of the increased disorder brought about by mixing. When two pure substances mix under normal conditions there is usually an increase in the entropy of the system. The Thomas Group - PTCL, Oxford Thermodynamics of Solutions - First Year, Trinity Term Entropy of Mixing 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
